These ratios by themselves may not seem particularly interesting or informative however, if we take a ratio of these ratios, we obtain a useful and possibly surprising result: a small, whole-number ratio. 
For example, copper and chlorine can form a green, crystalline solid with a mass ratio of 0.558 g chlorine to 1 g copper, as well as a brown crystalline solid with a mass ratio of 1.116 g chlorine to 1 g copper. The law of multiple proportions states that when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small, whole numbers. For example, there are many compounds other than isooctane that also have a carbon-to-hydrogen mass ratio of 5.33:1.00.ĭalton also used data from Proust, as well as results from his own experiments, to formulate another interesting law. 
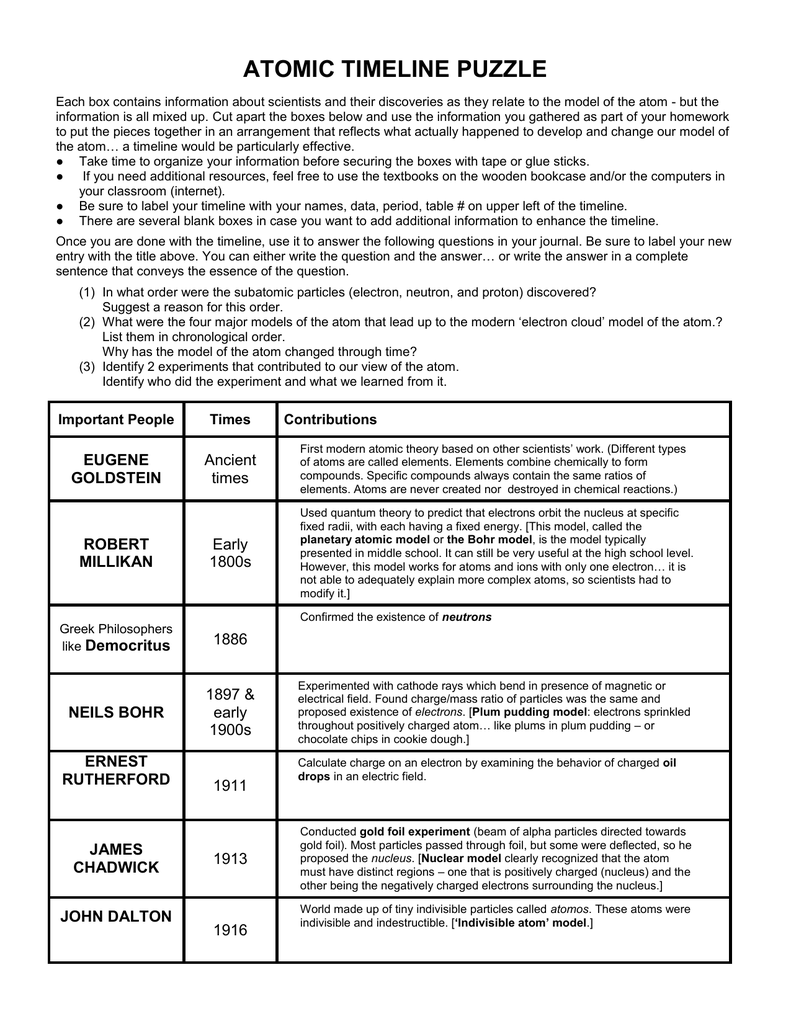
That is, samples that have the same mass ratio are not necessarily the same substance. It is worth noting that although all samples of a particular compound have the same mass ratio, the converse is not true in general. There is a page containing 8 multiple choice questions and the project rubric.\) Some students get creative by decorating with ways to remember the models: "chocolate chip cookie dough" around Thomson's information or "Legos" around Democritus'. I encourage them to make the poster colorful in order to separate the information that belongs together. The students read through the Information Book, cut out the "Everything You Need" pages and organize the images and information boxes into their own timeline. The Information Book is now also included as a digital Google document so you can share it with your students on Google Classroom and save paper! They end up being the perfect size to fit all of the information/pictures, and they look great hung up in my classroom and are not too big!Ĭopy one "Information Book" (it looks fine in Black and White - I copy it back-to-back, put the pages into four plastic sleeves, and bulldog clamp the pages for use each year) and one set of the Directions and "Everything You Need to Make an Atomic Theory Timeline" per group or pair of partners. I provide them with 11" by 17" white construction paper for the "posters". This journey begins well over 2000 years ago in Asia with categories of matter and continues through the Dark Ages where the quest for riches and everlasting life was sought through alchemy. I like to show this Ted Ed video before we begin: History of Atomic Theory Study Guide Created by Natalie Cielanga Discover the ideas that led to our current understanding of the struc-ture of the atom. (Empedocles) 494-530 BCE He theorized that everything is made of different combinations of Air, Earth, Water and Fire. The atoms have empty space between them, Atoms are indestructible and that their are infinite number of atoms. This project covers: Democritus, Aristotle and "the Death of Chemistry", Dalton, Thomson, Rutherford, Bohr, and the Modern Cloud Theory. He founder of the atomic theory, who theorized that everything is made of tiny particles called atoms. The Atomic Theory Timeline Project is everything you need to give your chemistry students a nice background on the history of the atom! Students use skills of synthesizing informational text, organizing pieces of information into groups and then into a chronological order, and displaying the information in a clear and creative way. Best Known For: Chemist John Dalton is credited with pioneering modern atomic theory.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
